Circle Pharma Announces Appointment of David Spellmeyer, PhD, as Chief Scientific Officer
SOUTH SAN FRANCISCO, Calif. – (BUSINESS WIRE) – Circle Pharma, Inc., a developer of macrocycle therapeutics, today announced that David Spellmeyer, PhD, has been appointed as its chief scientific officer.
“His deep experience in rational drug design and discovery is an excellent fit for Circle as we extend our platform for macrocycle discovery and advance our therapeutic pipeline towards the clinic.”
Dr. Spellmeyer brings over 25 years of broad industrial experience to Circle. He previously served as CTO & CIO at Nodality, CSO at Signature BioScience, Research Staff Member at IBM, as well as scientific, quality systems, and leadership roles at other biotechnology companies. David has been involved in more than 20 strategic corporate partnerships, M&A, and joint ventures, as well as several rounds of venture financing.
“We are delighted to welcome David to Circle’s senior leadership,” said David J. Earp, JD, PhD, Circle’s CEO. “His deep experience in rational drug design and discovery is an excellent fit for Circle as we extend our platform for macrocycle discovery and advance our therapeutic pipeline towards the clinic.”
“Circle is at an exciting point in its development. I am thrilled to be joining a company that has built such a promising discovery platform,” said Dr. Spellmeyer. “The combination of rational design and efficient, automated macrocycle synthesis is a new approach to this drug class, and the results so far are highly encouraging.”
Dr. Spellmeyer received his PhD in 1987 from UCLA, and completed his post-doctoral work in pharmaceutical chemistry at UCSF, where he holds an appointment as an adjunct associate professor in the Department of Pharmaceutical Chemistry. David was named as a Fellow of the American Chemical Society in 2009. He joins Circle’s team after working with the company in his role as an Executive-in-Residence at ShangPharma Innovation, an investor in Circle. He continues to advise ShangPharma in an EIR capacity.
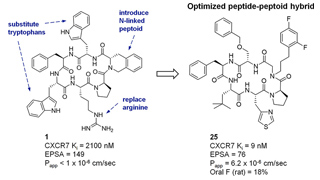
About Macrocyclic Peptides
Macrocyclic peptides have the potential to allow drug developers to address the large proportion of known therapeutic targets (estimated at up to 80%) that are considered undruggable with conventional small molecule or biologic modalities. In particular, there is great interest in developing macrocycles to modulate protein-protein interactions, which play a role in almost all disease conditions, including cancer, fibrosis, inflammation and infection. However, the development of macrocyclic therapeutics has been limited by the need for a greater understanding of how to develop macrocycles with appropriate pharmacokinetics, cell permeability and oral bioavailability. Circle is applying its ability to design potent macrocycles with intrinsic cell permeability and drug-like characteristics to unlock access to challenging, high value therapeutic targets that have been out of reach to other approaches.
About Circle Pharma, Inc.
Circle is developing a new paradigm for macrocycle drug discovery based on rational design and synthetic chemistry. Circle’s technology facilitates the design and synthesis of intrinsically cell-permeable macrocycles that can address both intra- and extra-cellular therapeutic targets, and can be delivered by oral administration. Circle’s macrocycle development platform is applicable across a wide range of serious diseases; the company is initially focusing its internal development efforts on intracellular protein-protein interactions that are key drivers in cancer. Circle’s founders are Prof. Matthew P. Jacobson (Chair of the Dept. of Pharmaceutical Chemistry at UC San Francisco and co-founder of Global Blood Therapeutics (NASDAQ: GBT) and Relay Therapeutics) and Prof. R. Scott Lokey (Dept. of Chemistry and Biochemistry, UC Santa Cruz and director of the UCSC Chemical Screening Center).
More information: www.circlepharma.com
Contacts
Circle Pharma, Inc.
Cayla McEwen, 650-392-0363
info@circlepharma.com
Read online article